Objectives After this lab you should be able to do the following: .
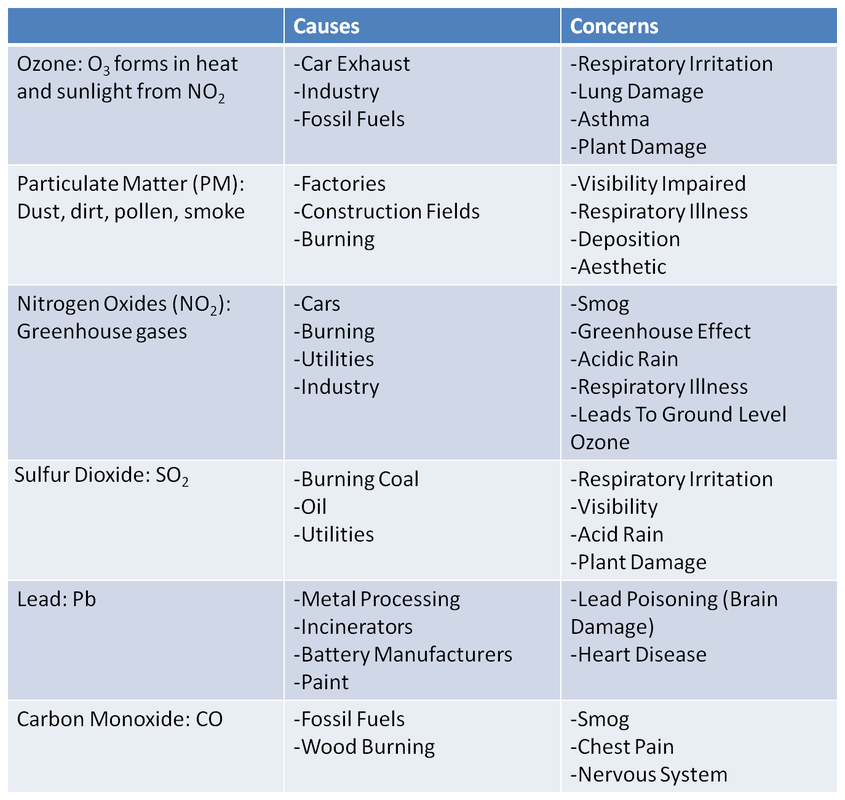
- Identify the six major air pollutants tested by the EPA.
- Understand different methods of testing air quality.
- Describe the air quality of Wake Forest using Ozone and PM measurements.
Materials Materials for this lab are:
Potassium Iodide
Corn starch
Beakers
Spatulas
Large Graduated Cylinder
Test Tube Rack
Filter Paper
Microscope slides
Petroleum Jelly
Q-tips
Microscopes
Problem What is the ozone level and PM level in Wake Forest?
Hypothesis The ozone level and PM level are both good in Wake Forest.
Introduction The Environmental Protection Agency name six air pollutants as Criteria Air Pollutants. These six are common in the United States and pose threats to health and the environment. The six are Ozone, Particulate Matter, Nitrogen Oxides, Sulfur Dioxide, Lead and Carbon Monoxide.
Ozone
Ozone (O3) is a gas composed of three oxygen atoms. It is not usually emitted directly into the air, but at ground level is created by a chemical reaction between oxides of nitrogen (NOx) and volatile organic compounds (VOC) in the presence of heat and sunlight. Ozone has the same chemical structure whether it occurs miles above the earth or at ground level and can be "good" or "bad," depending on its location in the atmosphere. "Good" ozone occurs naturally in the stratosphere approximately 10 to 30 miles above the earth's surface and forms a layer that protects life on earth from the sun's harmful rays. In the earth's lower atmosphere, ground-level ozone is considered "bad."
VOC + NOx + Heat + Sunlight = Ozone Motor vehicle exhaust and industrial emissions, gasoline vapors, and chemical solvents are some of the major sources of NOx and VOC, that help to form ozone. Sunlight and hot weather cause ground-level ozone to form in harmful concentrations in the air. As a result, it is known as a summertime air pollutant. Many urban areas tend to have high levels of "bad" ozone, but even rural areas are also subject to increased ozone levels because wind carries ozone and pollutants that form it hundreds of miles away from their original sources.
Health Problems -Ozone can irritate lung airways and cause inflammation much like a sunburn. Other symptoms include wheezing, coughing, pain when taking a deep breath, and breathing difficulties during exercise or outdoor activities. People with respiratory problems are most vulnerable, but even healthy people that are active outdoors can be affected when ozone levels are high.
Repeated exposure to ozone pollution for several months may cause permanent lung damage. Anyone who spends time outdoors in the summer is at risk, particularly children and other people who are active outdoors..
Even at very low levels, ground-level ozone triggers a variety of health problems including aggravated asthma, reduced lung capacity, and increased susceptibility to respiratory illnesses like pneumonia and bronchitis.
Plant and Ecosystem Damage - Ground-level ozone interferes with the ability of plants to produce and store food, which makes them more susceptible to disease, insects, other pollutants, and harsh weather.
Ozone damages the leaves of trees and other plants, ruining the appearance of cities, national parks, and recreation areas.
Ozone reduces crop and forest yields and increases plant vulnerability to disease, pests, and harsh weather.
Particulate Matter
Particulate matter, or PM, is the term for particles found in the air, including dust, dirt, soot, smoke, and liquid droplets. Particles can be suspended in the air for long periods of time. Some particles are large or dark enough to be seen as soot or smoke. Others are so small that individually they can only be detected with an electron microscope.
Some particles are directly emitted into the air. They come from a variety of sources such as cars, trucks, buses, factories, construction sites, tilled fields, unpaved roads, stone crushing, and burning of wood.
Health effects - Many scientific studies have linked breathing PM to a series of significant health problems, including: aggravated asthma, increases in respiratory symptoms like coughing and difficult or painful breathing, chronic bronchitis, decreased lung function, and premature death
Visibility impairment - PM is the major cause of reduced visibility (haze) in parts of the United States, including many of our national parks.
Atmospheric deposition - Particles can be carried over long distances by wind and then settle on ground or water. The effects of this settling include: making lakes and streams acidic, changing the nutrient balance in coastal waters and large river basins, depleting the nutrients in soil, damaging sensitive forests and farm crops, and affecting the diversity of ecosystems
Aesthetic damage - Soot, a type of PM, stains and damages stone and other materials, including culturally important objects such as monuments and statues.
Sulfur Dioxide Sulfur dioxide, or SO2, belongs to the family of sulfur oxide gases (SOx). These gases dissolve easily in water. Sulfur is prevalent in all raw materials, including crude oil, coal, and ore that contains common metals like aluminum, copper, zinc, lead, and iron. SOx gases are formed when fuel containing sulfur, such as coal and oil, is burned, and when gasoline is extracted from oil, or metals are extracted from ore. SO2 dissolves in water vapor to form acid, and interacts with other gases and particles in the air to form sulfates and other products that can be harmful to people and their environment.
Over 65% of SO2 released to the air, or more than 13 million tons per year, comes from electric utilities, especially those that burn coal. Other sources of SO2 are industrial facilities that derive their products from raw materials like metallic ore, coal, and crude oil, or that burn coal or oil to produce process heat. Examples are petroleum refineries, cement manufacturing, and metal processing facilities. Also, locomotives, large ships, and some non-road diesel equipment currently burn high sulfur fuel and release SO2 emissions to the air in large quantities.
Respiratory Effects from Gaseous SO2 - Peak levels of SO2 in the air can cause temporary breathing difficulty for people with asthma who are active outdoors. Longer-term exposures to high levels of SO2 gas and particles cause respiratory illness and aggravate existing heart disease.
Respiratory Effects from Sulfate Particles - SO2 reacts with other chemicals in the air to form tiny sulfate particles. When these are breathed, they gather in the lungs and are associated with increased respiratory symptoms and disease, difficulty in breathing, and premature death.
Visibility Impairment - Haze occurs when light is scattered or absorbed by particles and gases in the air. Sulfate particles are the major cause of reduced visibility in many parts of the U.S., including our national parks.
Acid Rain - SO2 and nitrogen oxides react with other substances in the air to form acids, which fall to earth as rain, fog, snow, or dry particles. Some may be carried by the wind for hundreds of miles.
Plant and Water Damage - Acid rain damages forests and crops, changes the makeup of soil, and makes lakes and streams acidic and unsuitable for fish. Continued exposure over a long time changes the natural variety of plants and animals in an ecosystem.
Aesthetic Damage - SO2 accelerates the decay of building materials and paints, including irreplaceable monuments, statues, and sculptures that are part of our nation's cultural heritage.
Nitrogen Oxides Nitrogen oxides, or NOx, is the generic term for a group of highly reactive gases, all of which contain nitrogen and oxygen in varying amounts. Many of the nitrogen oxides are colorless and odorless. However, one common pollutant, nitrogen dioxide (NO2) along with particles in the air can often be seen as a reddish-brown layer over many urban areas.
Nitrogen oxides form when fuel is burned at high temperatures, as in a combustion process. The primary sources of NOx are motor vehicles, electric utilities, and other industrial, commercial, and residential sources that burn fuels.
Ground-level Ozone (Smog) - is formed when NOx and volatile organic compounds (VOCs) react in the presence of heat and sunlight. Children, people with lung diseases such as asthma, and people who work or exercise outside are susceptible to adverse effects such as damage to lung tissue and reduction in lung function. Ozone can be transported by wind currents and cause health impacts far from original sources. Millions of Americans live in areas that do not meet the health standards for ozone. Other impacts from ozone include damaged vegetation and reduced crop yields.
Acid Rain - NOx and sulfur dioxide react with other substances in the air to form acids which fall to earth as rain, fog, snow or dry particles. Some may be carried by wind for hundreds of miles. Acid rain damages; causes deterioration of cars, buildings and historical monuments; and causes lakes and streams to become acidic and unsuitable for many fish.
Particles - NOx reacts with ammonia, moisture, and other compounds to form nitric acid and related particles. Human health concerns include effects on breathing and the respiratory system, damage to lung tissue, and premature death. Small particles penetrate deeply into sensitive parts of the lungs and can cause or worsen respiratory disease such as emphysema and bronchitis, and aggravate existing heart disease.
Water Quality Deterioration - Increased nitrogen loading in water bodies, particularly coastal estuaries, upsets the chemical balance of nutrients used by aquatic plants and animals. Additional nitrogen accelerates "eutrophication," which leads to oxygen depletion and reduces fish and shellfish populations. NOx emissions in the air are one of the largest sources of nitrogen pollution in the Chesapeake Bay.
Global Warming - One member of the NOx, nitrous oxide, is a greenhouse gas. It accumulates in the atmosphere with other greenhouse gasses causing a gradual rise in the earth's temperature. This will lead to increased risks to human health, a rise in the sea level, and other adverse changes to plant and animal habitat.
Toxic Chemicals - In the air, NOx reacts readily with common organic chemicals and even ozone, to form a wide variety of toxic products, some of which may cause biological mutations. Examples of these chemicals include the nitrate radical, nitroarenes, and nitrosamines.
Visibility Impairment - Nitrate particles and nitrogen dioxide can block the transmission of light, reducing visibility in urban areas and on a regional scale in our national parks.
Lead Lead is a metal found naturally in the environment as well as in manufactured products. The major sources of lead emissions have historically been motor vehicles (such as cars and trucks) and industrial sources. Due to the phase out of leaded gasoline, metals processing is the major source of lead emissions to the air today. The highest levels of lead in air are generally found near lead smelters. Other stationary sources are waste incinerators, utilities, and lead-acid battery manufacturers.
Damages organs - Lead causes damage to the kidneys, liver, brain and nerves, and other organs. Exposure to lead may also lead to osteoporosis (brittle bone disease) and reproductive disorders.
Affects the brain and nerves - Excessive exposure to lead causes seizures, mental retardation, behavioral disorders, memory problems, and mood changes. Low levels of lead damage the brain and nerves in fetuses and young children, resulting in learning deficits and lowered IQ.
Affects the heart and blood - Lead exposure causes high blood pressure and increases heart disease, especially in men. Lead exposure may also lead to anemia, or weak blood.
Affects animals and plants - Wild and domestic animals can ingest lead while grazing. They experience the same kind of effects as people who are exposed to lead. Low concentrations of lead can slow down vegetation growth near industrial facilities
Affects fish - Lead can enter water systems through runoff and from sewage and industrial waste streams. Elevated levels of lead in the water can cause reproductive damage in some aquatic life and cause blood and neurological changes in fish and other animals that live there.
Carbon Monoxide Carbon monoxide, or CO, is a colorless, odorless gas that is formed when carbon in fuel is not burned completely. It is a component of motor vehicle exhaust, which contributes about 56 percent of all CO emissions nationwide. Other non-road engines and vehicles (such as construction equipment and boats) contribute about 22 percent of all CO emissions nationwide. Higher levels of CO generally occur in areas with heavy traffic congestion. In cities, 85 to 95 percent of all CO emissions may come from motor vehicle exhaust. Other sources of CO emissions include industrial processes (such as metals processing and chemical manufacturing), residential wood burning, and natural sources such as forest fires. Woodstoves, gas stoves, cigarette smoke, and unvented gas and kerosene space heaters are sources of CO indoors. The highest levels of CO in the outside air typically occur during the colder months of the year when inversion conditions are more frequent. The air pollution becomes trapped near the ground beneath a layer of warm air.
Cardiovascular Effects - The health threat from lower levels of CO is most serious for those who suffer from heart disease, like angina, clogged arteries, or congestive heart failure. For a person with heart disease, a single exposure to CO at low levels may cause chest pain and reduce that person's ability to exercise; repeated exposures may contribute to other cardiovascular effects.
Central Nervous System Effects - Even healthy people can be affected by high levels of CO. People who breathe high levels of CO can develop vision problems, reduced ability to work or learn, reduced manual dexterity, and difficulty performing complex tasks. At extremely high levels, CO is poisonous and can cause death.
Smog - CO contributes to the formation of smog ground-level ozone, which can trigger serious respiratory problems.
Pre-lab Questions 1. Create a chart showing the six air pollutants, causes, and concerns from the information in the pre-lab.
- Identify the six major air pollutants tested by the EPA.
- Understand different methods of testing air quality.
- Describe the air quality of Wake Forest using Ozone and PM measurements.
Materials Materials for this lab are:
Potassium Iodide
Corn starch
Beakers
Spatulas
Large Graduated Cylinder
Test Tube Rack
Filter Paper
Microscope slides
Petroleum Jelly
Q-tips
Microscopes
Problem What is the ozone level and PM level in Wake Forest?
Hypothesis The ozone level and PM level are both good in Wake Forest.
Introduction The Environmental Protection Agency name six air pollutants as Criteria Air Pollutants. These six are common in the United States and pose threats to health and the environment. The six are Ozone, Particulate Matter, Nitrogen Oxides, Sulfur Dioxide, Lead and Carbon Monoxide.
Ozone
Ozone (O3) is a gas composed of three oxygen atoms. It is not usually emitted directly into the air, but at ground level is created by a chemical reaction between oxides of nitrogen (NOx) and volatile organic compounds (VOC) in the presence of heat and sunlight. Ozone has the same chemical structure whether it occurs miles above the earth or at ground level and can be "good" or "bad," depending on its location in the atmosphere. "Good" ozone occurs naturally in the stratosphere approximately 10 to 30 miles above the earth's surface and forms a layer that protects life on earth from the sun's harmful rays. In the earth's lower atmosphere, ground-level ozone is considered "bad."
VOC + NOx + Heat + Sunlight = Ozone Motor vehicle exhaust and industrial emissions, gasoline vapors, and chemical solvents are some of the major sources of NOx and VOC, that help to form ozone. Sunlight and hot weather cause ground-level ozone to form in harmful concentrations in the air. As a result, it is known as a summertime air pollutant. Many urban areas tend to have high levels of "bad" ozone, but even rural areas are also subject to increased ozone levels because wind carries ozone and pollutants that form it hundreds of miles away from their original sources.
Health Problems -Ozone can irritate lung airways and cause inflammation much like a sunburn. Other symptoms include wheezing, coughing, pain when taking a deep breath, and breathing difficulties during exercise or outdoor activities. People with respiratory problems are most vulnerable, but even healthy people that are active outdoors can be affected when ozone levels are high.
Repeated exposure to ozone pollution for several months may cause permanent lung damage. Anyone who spends time outdoors in the summer is at risk, particularly children and other people who are active outdoors..
Even at very low levels, ground-level ozone triggers a variety of health problems including aggravated asthma, reduced lung capacity, and increased susceptibility to respiratory illnesses like pneumonia and bronchitis.
Plant and Ecosystem Damage - Ground-level ozone interferes with the ability of plants to produce and store food, which makes them more susceptible to disease, insects, other pollutants, and harsh weather.
Ozone damages the leaves of trees and other plants, ruining the appearance of cities, national parks, and recreation areas.
Ozone reduces crop and forest yields and increases plant vulnerability to disease, pests, and harsh weather.
Particulate Matter
Particulate matter, or PM, is the term for particles found in the air, including dust, dirt, soot, smoke, and liquid droplets. Particles can be suspended in the air for long periods of time. Some particles are large or dark enough to be seen as soot or smoke. Others are so small that individually they can only be detected with an electron microscope.
Some particles are directly emitted into the air. They come from a variety of sources such as cars, trucks, buses, factories, construction sites, tilled fields, unpaved roads, stone crushing, and burning of wood.
Health effects - Many scientific studies have linked breathing PM to a series of significant health problems, including: aggravated asthma, increases in respiratory symptoms like coughing and difficult or painful breathing, chronic bronchitis, decreased lung function, and premature death
Visibility impairment - PM is the major cause of reduced visibility (haze) in parts of the United States, including many of our national parks.
Atmospheric deposition - Particles can be carried over long distances by wind and then settle on ground or water. The effects of this settling include: making lakes and streams acidic, changing the nutrient balance in coastal waters and large river basins, depleting the nutrients in soil, damaging sensitive forests and farm crops, and affecting the diversity of ecosystems
Aesthetic damage - Soot, a type of PM, stains and damages stone and other materials, including culturally important objects such as monuments and statues.
Sulfur Dioxide Sulfur dioxide, or SO2, belongs to the family of sulfur oxide gases (SOx). These gases dissolve easily in water. Sulfur is prevalent in all raw materials, including crude oil, coal, and ore that contains common metals like aluminum, copper, zinc, lead, and iron. SOx gases are formed when fuel containing sulfur, such as coal and oil, is burned, and when gasoline is extracted from oil, or metals are extracted from ore. SO2 dissolves in water vapor to form acid, and interacts with other gases and particles in the air to form sulfates and other products that can be harmful to people and their environment.
Over 65% of SO2 released to the air, or more than 13 million tons per year, comes from electric utilities, especially those that burn coal. Other sources of SO2 are industrial facilities that derive their products from raw materials like metallic ore, coal, and crude oil, or that burn coal or oil to produce process heat. Examples are petroleum refineries, cement manufacturing, and metal processing facilities. Also, locomotives, large ships, and some non-road diesel equipment currently burn high sulfur fuel and release SO2 emissions to the air in large quantities.
Respiratory Effects from Gaseous SO2 - Peak levels of SO2 in the air can cause temporary breathing difficulty for people with asthma who are active outdoors. Longer-term exposures to high levels of SO2 gas and particles cause respiratory illness and aggravate existing heart disease.
Respiratory Effects from Sulfate Particles - SO2 reacts with other chemicals in the air to form tiny sulfate particles. When these are breathed, they gather in the lungs and are associated with increased respiratory symptoms and disease, difficulty in breathing, and premature death.
Visibility Impairment - Haze occurs when light is scattered or absorbed by particles and gases in the air. Sulfate particles are the major cause of reduced visibility in many parts of the U.S., including our national parks.
Acid Rain - SO2 and nitrogen oxides react with other substances in the air to form acids, which fall to earth as rain, fog, snow, or dry particles. Some may be carried by the wind for hundreds of miles.
Plant and Water Damage - Acid rain damages forests and crops, changes the makeup of soil, and makes lakes and streams acidic and unsuitable for fish. Continued exposure over a long time changes the natural variety of plants and animals in an ecosystem.
Aesthetic Damage - SO2 accelerates the decay of building materials and paints, including irreplaceable monuments, statues, and sculptures that are part of our nation's cultural heritage.
Nitrogen Oxides Nitrogen oxides, or NOx, is the generic term for a group of highly reactive gases, all of which contain nitrogen and oxygen in varying amounts. Many of the nitrogen oxides are colorless and odorless. However, one common pollutant, nitrogen dioxide (NO2) along with particles in the air can often be seen as a reddish-brown layer over many urban areas.
Nitrogen oxides form when fuel is burned at high temperatures, as in a combustion process. The primary sources of NOx are motor vehicles, electric utilities, and other industrial, commercial, and residential sources that burn fuels.
Ground-level Ozone (Smog) - is formed when NOx and volatile organic compounds (VOCs) react in the presence of heat and sunlight. Children, people with lung diseases such as asthma, and people who work or exercise outside are susceptible to adverse effects such as damage to lung tissue and reduction in lung function. Ozone can be transported by wind currents and cause health impacts far from original sources. Millions of Americans live in areas that do not meet the health standards for ozone. Other impacts from ozone include damaged vegetation and reduced crop yields.
Acid Rain - NOx and sulfur dioxide react with other substances in the air to form acids which fall to earth as rain, fog, snow or dry particles. Some may be carried by wind for hundreds of miles. Acid rain damages; causes deterioration of cars, buildings and historical monuments; and causes lakes and streams to become acidic and unsuitable for many fish.
Particles - NOx reacts with ammonia, moisture, and other compounds to form nitric acid and related particles. Human health concerns include effects on breathing and the respiratory system, damage to lung tissue, and premature death. Small particles penetrate deeply into sensitive parts of the lungs and can cause or worsen respiratory disease such as emphysema and bronchitis, and aggravate existing heart disease.
Water Quality Deterioration - Increased nitrogen loading in water bodies, particularly coastal estuaries, upsets the chemical balance of nutrients used by aquatic plants and animals. Additional nitrogen accelerates "eutrophication," which leads to oxygen depletion and reduces fish and shellfish populations. NOx emissions in the air are one of the largest sources of nitrogen pollution in the Chesapeake Bay.
Global Warming - One member of the NOx, nitrous oxide, is a greenhouse gas. It accumulates in the atmosphere with other greenhouse gasses causing a gradual rise in the earth's temperature. This will lead to increased risks to human health, a rise in the sea level, and other adverse changes to plant and animal habitat.
Toxic Chemicals - In the air, NOx reacts readily with common organic chemicals and even ozone, to form a wide variety of toxic products, some of which may cause biological mutations. Examples of these chemicals include the nitrate radical, nitroarenes, and nitrosamines.
Visibility Impairment - Nitrate particles and nitrogen dioxide can block the transmission of light, reducing visibility in urban areas and on a regional scale in our national parks.
Lead Lead is a metal found naturally in the environment as well as in manufactured products. The major sources of lead emissions have historically been motor vehicles (such as cars and trucks) and industrial sources. Due to the phase out of leaded gasoline, metals processing is the major source of lead emissions to the air today. The highest levels of lead in air are generally found near lead smelters. Other stationary sources are waste incinerators, utilities, and lead-acid battery manufacturers.
Damages organs - Lead causes damage to the kidneys, liver, brain and nerves, and other organs. Exposure to lead may also lead to osteoporosis (brittle bone disease) and reproductive disorders.
Affects the brain and nerves - Excessive exposure to lead causes seizures, mental retardation, behavioral disorders, memory problems, and mood changes. Low levels of lead damage the brain and nerves in fetuses and young children, resulting in learning deficits and lowered IQ.
Affects the heart and blood - Lead exposure causes high blood pressure and increases heart disease, especially in men. Lead exposure may also lead to anemia, or weak blood.
Affects animals and plants - Wild and domestic animals can ingest lead while grazing. They experience the same kind of effects as people who are exposed to lead. Low concentrations of lead can slow down vegetation growth near industrial facilities
Affects fish - Lead can enter water systems through runoff and from sewage and industrial waste streams. Elevated levels of lead in the water can cause reproductive damage in some aquatic life and cause blood and neurological changes in fish and other animals that live there.
Carbon Monoxide Carbon monoxide, or CO, is a colorless, odorless gas that is formed when carbon in fuel is not burned completely. It is a component of motor vehicle exhaust, which contributes about 56 percent of all CO emissions nationwide. Other non-road engines and vehicles (such as construction equipment and boats) contribute about 22 percent of all CO emissions nationwide. Higher levels of CO generally occur in areas with heavy traffic congestion. In cities, 85 to 95 percent of all CO emissions may come from motor vehicle exhaust. Other sources of CO emissions include industrial processes (such as metals processing and chemical manufacturing), residential wood burning, and natural sources such as forest fires. Woodstoves, gas stoves, cigarette smoke, and unvented gas and kerosene space heaters are sources of CO indoors. The highest levels of CO in the outside air typically occur during the colder months of the year when inversion conditions are more frequent. The air pollution becomes trapped near the ground beneath a layer of warm air.
Cardiovascular Effects - The health threat from lower levels of CO is most serious for those who suffer from heart disease, like angina, clogged arteries, or congestive heart failure. For a person with heart disease, a single exposure to CO at low levels may cause chest pain and reduce that person's ability to exercise; repeated exposures may contribute to other cardiovascular effects.
Central Nervous System Effects - Even healthy people can be affected by high levels of CO. People who breathe high levels of CO can develop vision problems, reduced ability to work or learn, reduced manual dexterity, and difficulty performing complex tasks. At extremely high levels, CO is poisonous and can cause death.
Smog - CO contributes to the formation of smog ground-level ozone, which can trigger serious respiratory problems.
Pre-lab Questions 1. Create a chart showing the six air pollutants, causes, and concerns from the information in the pre-lab.
2. Why is ground level ozone highest in the summer and almost nonexistent in the winter? How might this affect our results as we test the air during this part of the year?
· During the summer there is more sunlight, therefore, more heat which creates more ozone. On the contrary, cold temperatures don’t produce this effect. You would get an inaccurate temperature.
3. Many third world countries still use leaded gasoline (since they have older cars or since they have not forced changes). What concerns may the governments of these countries need to deal with?
· They might deal with health problems like the damage of people’s organs, brain and nerve effects, and the effect on plants and animals.
4. Use the internet to find what parts of North Carolina are not in compliance with the Standards of the Clean Air Act. You may give either general areas or specific counties.
· Central region of North Carolina.
Procedure Preparation 1. Half the class should work on preparing the ozone monitoring test strips by placing 100ml of water in a 250 ml beaker, and adding 5g of cornstarch. Then heat and stir the mixture until it gels (when the mixture becomes thick and clear). Remove the solution from heat, add 1 g of potassium iodide, stir well, and allow to cool. After the solution has cooled, lay a piece of filter paper on a petri and brush the paste onto the filter paper. Turn the filter paper and brush the paste on the other side. Hang up and allow the filter paper to dry. Cut the papers into 1 inch-wide strips. Store the strips in a plastic bag out of sunlight.
2. The other half of the class will prepare Particulate matter collectors by smearing a thin layer of petroleum jelly onto a microscope slides. Try to make the layer as even as possible. Use a q-tip to lightly smear the specimen side of each slide. You will need to prepare 2 slides per group and place them in a slide container to avoid smearing the petroleum jelly on anyone.
Data Collection 3. Each group will place one labeled slide on the exterior of the school. Some groups should choose areas that would receive few particulates, others should choose locations that produce more particulates. (Keep in mind sources of particulate matter)
4. One member from each group should take the second slide and place it in a location inside the school.
5. You should then place your ozone monitoring strip outside for 8 hrs. Before placing the strip outside, dip the strip in distilled water, then attach the strip out of sunlight. When placing your strip find the relative humidity for that day.
6. The next day, each group should collect their slide and ozone monitoring strip.
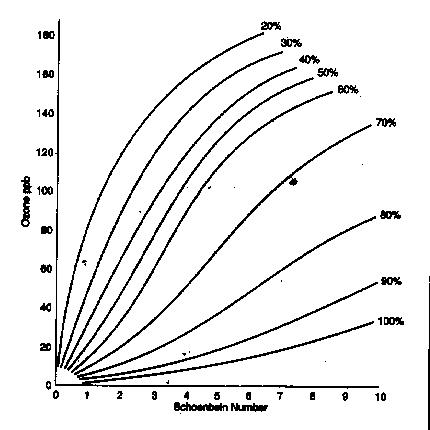
Schoenbein Number
6. Dunk each strip into distilled water. Use the chart at the right to determine the Schoenbein Number of your strip.
Christian Friedrich Schoenbein discovered ozone in 1839 during his tenure as a professor at the University of Basel, Switzerland. He used the reactivity of ozone to measure its presence and demonstrate that it is a naturally occurring component of the atmosphere. He developed a way to measure ozone in the troposphere using a mixture of starch, potassium iodide, and water spread on filter paper. The paper, called Schoenbein paper, changes color when ozone is present. Ozone causes iodide to oxidize into iodine ( ).
This test is based on the oxidation capability of ozone. Ozone in the air will oxidize the potassium iodide on the test paper to produce iodine. The iodine reacts with starch, staining the paper a shade of purple. The intensity of the purple color depends on the amount of ozone present in the air. The darker the color, the more ozone is present.
The reactions involved are:
2KI + O3 + H2O --> 2KOH + O2 + I2
I2 + starch --> blue color
7. Use the relative humidity and the Schoenbein number to calculate the concentration of ground level ozone using the chart below.
· During the summer there is more sunlight, therefore, more heat which creates more ozone. On the contrary, cold temperatures don’t produce this effect. You would get an inaccurate temperature.
3. Many third world countries still use leaded gasoline (since they have older cars or since they have not forced changes). What concerns may the governments of these countries need to deal with?
· They might deal with health problems like the damage of people’s organs, brain and nerve effects, and the effect on plants and animals.
4. Use the internet to find what parts of North Carolina are not in compliance with the Standards of the Clean Air Act. You may give either general areas or specific counties.
· Central region of North Carolina.
Procedure Preparation 1. Half the class should work on preparing the ozone monitoring test strips by placing 100ml of water in a 250 ml beaker, and adding 5g of cornstarch. Then heat and stir the mixture until it gels (when the mixture becomes thick and clear). Remove the solution from heat, add 1 g of potassium iodide, stir well, and allow to cool. After the solution has cooled, lay a piece of filter paper on a petri and brush the paste onto the filter paper. Turn the filter paper and brush the paste on the other side. Hang up and allow the filter paper to dry. Cut the papers into 1 inch-wide strips. Store the strips in a plastic bag out of sunlight.
2. The other half of the class will prepare Particulate matter collectors by smearing a thin layer of petroleum jelly onto a microscope slides. Try to make the layer as even as possible. Use a q-tip to lightly smear the specimen side of each slide. You will need to prepare 2 slides per group and place them in a slide container to avoid smearing the petroleum jelly on anyone.
Data Collection 3. Each group will place one labeled slide on the exterior of the school. Some groups should choose areas that would receive few particulates, others should choose locations that produce more particulates. (Keep in mind sources of particulate matter)
4. One member from each group should take the second slide and place it in a location inside the school.
5. You should then place your ozone monitoring strip outside for 8 hrs. Before placing the strip outside, dip the strip in distilled water, then attach the strip out of sunlight. When placing your strip find the relative humidity for that day.
6. The next day, each group should collect their slide and ozone monitoring strip.
Schoenbein Number
6. Dunk each strip into distilled water. Use the chart at the right to determine the Schoenbein Number of your strip.
Christian Friedrich Schoenbein discovered ozone in 1839 during his tenure as a professor at the University of Basel, Switzerland. He used the reactivity of ozone to measure its presence and demonstrate that it is a naturally occurring component of the atmosphere. He developed a way to measure ozone in the troposphere using a mixture of starch, potassium iodide, and water spread on filter paper. The paper, called Schoenbein paper, changes color when ozone is present. Ozone causes iodide to oxidize into iodine ( ).
This test is based on the oxidation capability of ozone. Ozone in the air will oxidize the potassium iodide on the test paper to produce iodine. The iodine reacts with starch, staining the paper a shade of purple. The intensity of the purple color depends on the amount of ozone present in the air. The darker the color, the more ozone is present.
The reactions involved are:
2KI + O3 + H2O --> 2KOH + O2 + I2
I2 + starch --> blue color
7. Use the relative humidity and the Schoenbein number to calculate the concentration of ground level ozone using the chart below.
8. Look at your particulate matter slide under the microscope. First decide the amount of particulates and then attempt to estimate the source of the PM. Your group will need to decide how you can classify what type of PM was collected.
9. Make sure you record all you lab information into your notebook.
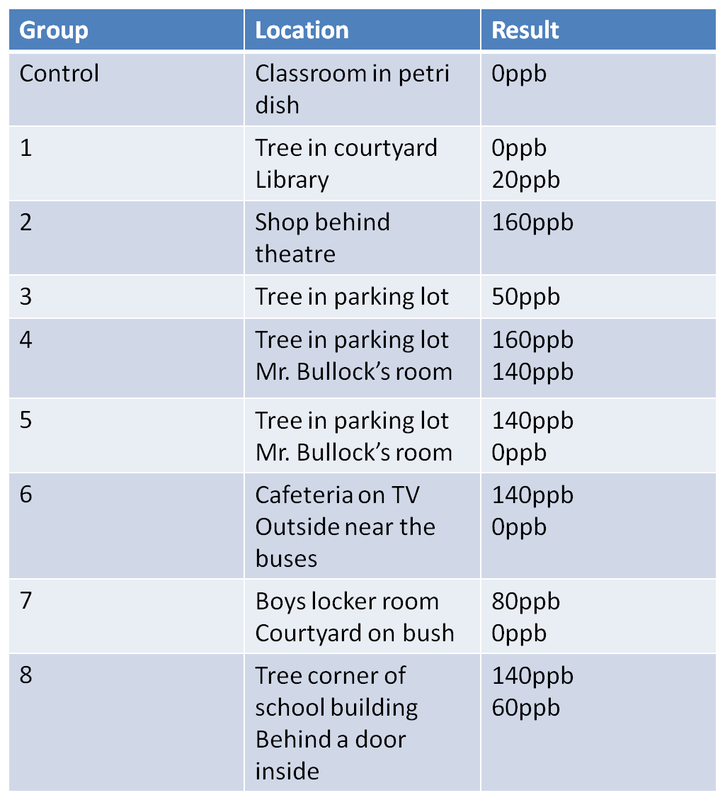
OZONE RESULTS
9. Make sure you record all you lab information into your notebook.
OZONE RESULTS
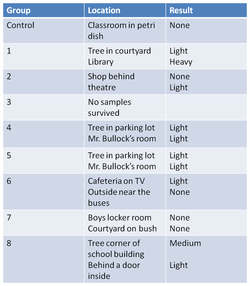
PM RESULTS
Data collected 9/7/12-9/10/12
Weather: Sunny on Friday, Sunday, and Monday, Rain on Saturday
Temperature on 9/10/12: 22°C
Relative Humidity on 9/10/12: 65%
Lab Questions 1. The highest rating the EPA will allow for a community for ground level ozone is 80 ppb. Any community with over 80 ppb for an average over three years is in noncompliance with the Clean Air Act. Compare your number to this standard. Did Wake Forest meet the standard on this day? Using what you know about the formation of ozone, decide if you believe the measurement you made was an accurate measurement of the air quality and why.
· My group was group 7 and we did meet the standards, but other groups did not meet the standards. Overall, Wake Forest did not meet the CAA standards for the day. The measurement that was made was accurate because everything was done the way it had to be done.
2. 13% of counties in the US are not meeting the Ozone requirements of the Clean Air Act. Use the information from class to explain the reason for the location of the counties in Non-attainment.
· The reason of these counties are considered non-attainment is because they are in areas where ozone is very high because of fossil fuels going up to the troposphere.
3. Particulate matter has recently been added to the requirements of the Clean Air Act. Theorize reasons that it was not part of the act until the 1990’s.
· Reasons as to why PM was not added to the requirements of the CAA are because PM was not a big deal before the 1990’s and it probably wasn’t even known to people except for scientists.
4. Compare and contrast the different PM samples – did they meet your expectations? Why or why not?
· Most of them met my expectations because most of them only had light PM.
5. Explain why air quality monitoring is important for the health of the economy.
· It is important for the health of the economy because then a lot of our ozone would be bad and there would be many cases of respiratory issues.
Independent Variable: Location
Dependent Variable: Ozone and PM level
Controlled Variables: Potassium Iodide and Petroleum Jelly
Control Group: Potassium Iodide and Petroleum Jelly
Experimental Group: Potassium Iodide and Petroleum Jelly
My hypothesis was half correct. The PM levels were good, but the ozone level was in the middle of good and bad.
Weather: Sunny on Friday, Sunday, and Monday, Rain on Saturday
Temperature on 9/10/12: 22°C
Relative Humidity on 9/10/12: 65%
Lab Questions 1. The highest rating the EPA will allow for a community for ground level ozone is 80 ppb. Any community with over 80 ppb for an average over three years is in noncompliance with the Clean Air Act. Compare your number to this standard. Did Wake Forest meet the standard on this day? Using what you know about the formation of ozone, decide if you believe the measurement you made was an accurate measurement of the air quality and why.
· My group was group 7 and we did meet the standards, but other groups did not meet the standards. Overall, Wake Forest did not meet the CAA standards for the day. The measurement that was made was accurate because everything was done the way it had to be done.
2. 13% of counties in the US are not meeting the Ozone requirements of the Clean Air Act. Use the information from class to explain the reason for the location of the counties in Non-attainment.
· The reason of these counties are considered non-attainment is because they are in areas where ozone is very high because of fossil fuels going up to the troposphere.
3. Particulate matter has recently been added to the requirements of the Clean Air Act. Theorize reasons that it was not part of the act until the 1990’s.
· Reasons as to why PM was not added to the requirements of the CAA are because PM was not a big deal before the 1990’s and it probably wasn’t even known to people except for scientists.
4. Compare and contrast the different PM samples – did they meet your expectations? Why or why not?
· Most of them met my expectations because most of them only had light PM.
5. Explain why air quality monitoring is important for the health of the economy.
· It is important for the health of the economy because then a lot of our ozone would be bad and there would be many cases of respiratory issues.
Independent Variable: Location
Dependent Variable: Ozone and PM level
Controlled Variables: Potassium Iodide and Petroleum Jelly
Control Group: Potassium Iodide and Petroleum Jelly
Experimental Group: Potassium Iodide and Petroleum Jelly
My hypothesis was half correct. The PM levels were good, but the ozone level was in the middle of good and bad.